Did you know that some amphibians and fish have hearts that regenerate muscular tissue cells following injury? This healing mechanism, called cardiac regeneration, is sustained throughout the creatures’ lives.
But for humans and other mammals, the heart’s reparative ability is confined to a small window of time during infancy. For mice, studies have shown that this window of time is about seven days. The heart of an infant mouse will self-repair in response to damage during this period, after which the capacity for cardiac regeneration is lost.

Sylvie Cohen (Far Left) with the other vet students that participated in the NIH internship program and the two directors of the program (Dr. Mark Simpson, DVM, PhD and Dr. Chuck Halsey, DVM, PhD).
Sylvie Cohen, second-year student at Ohio State’s College of Veterinary Medicine closely examined cardiac regeneration processes in one-day-old mice this summer at the National Heart, Lung and Blood Institute’s Laboratory of Molecular Biology. Cohen’s focus was on studying the molecular and cellular channels underlying heart-regeneration in mice.
Cohen and her team worked in the lab of Dr. Toren Finkel, senior investigator at the NIH’s Laboratory of Molecular Biology. First, the team wanted to test whether the infant mice’s hearts would actually repair themselves following injury. To do this, a microsurgery was performed on the experimental group that involved cutting off blood supply to the left anterior descending coronary artery, inducing a heart attack (modeled in figure below).
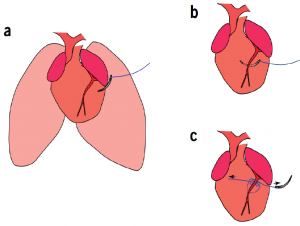
To establish how the neonatal mouse heart responds to ischemic injury, Cohen and her team permanently ligated the left anterior descending coronary artery of 1-day-old mice.
After 21 days, the mice that had heart attacks had fully recovered as their hearts had gradually generated new muscular tissue. Cohen and her team were able to see the cardiac regeneration process in action through histologic analyses of the mice’s heart tissue. As seen in the figure to the left the heart increasingly repaired itself over time, getting back to normal around day 21.
In addition to testing the mammalian heart’s capacity to regenerate for a brief period after birth, Cohen and her team also looked at what they consider a key-regulator in the cardiac regeneration process: a gene called BMI1. BMI1 has been shown to play an important role in cardiac muscle development, so researchers have hypothesized that it’s vital during cardiac regeneration.
To measure BMI1’s effect on heart regeneration, Cohen’s team compared the rate of cardiac healing in normal infant mice against BMI1-knockout infant mice. They found that the mice without the BMI1 gene had a reduced ability to renew cardiac muscle tissue, suggesting that BMI1 “is an important regulator of cardiac regeneration,” Cohen said.
Heart disease is the leading cause of death in the U.S., claiming the lives of more than 600,000 people per year, according to the Centers for Disease Control and Prevention. “Identifying key regulators of the cardiac regeneration process is an important step toward potential regenerative therapies such as gene therapy or pharmaceuticals” Cohen said.
Working with doctors and students from both veterinary and human medicine really enhanced the team’s collaboration and Cohen’s understanding of the research, she said. “It helped us maintain a holistic view.”
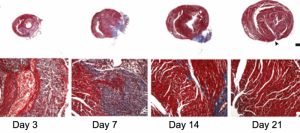
To assess the regenerative capacity of the neonatal mouse heart after ischemic injury, histological analyses were performed after coronary artery ligation. By 21 days after myocardial infarction, there was little evidence of fibrosis when using Masson’s trichrome staining.
This study was a part of the NIH’s Summer Internship Program in Biomedical Research for Veterinary Medical Students.
Comments are closed